posted
On Tuesday, October 29, 2019
in
Blog
 Steel has been around for ages, even the Romans used it back in 223 B.C. Ancient civilizations were able to find a way to make steel, as almost 5% of the earth’s crust consists of iron, making it the second most abundant metal on earth.[1]
Steel has been around for ages, even the Romans used it back in 223 B.C. Ancient civilizations were able to find a way to make steel, as almost 5% of the earth’s crust consists of iron, making it the second most abundant metal on earth.[1]
Steel is made through the addition of carbon into iron (up to 2%) which happens with extremely high heat. One part of steel production is heat treatment. Heat treatment is a method used to make metals stronger, harder and more durable. This method is very important to many steel and metallic parts.
Heat Treatment Benefits
Heat Treating of steel and other metals can lead to:
- Improved wear resistance
- Increased resistance to deformation and warpage and
- Increased strength or toughness
How Heat Treatment Works
When common metals, such as steels, are heated to high temperatures, there is a significant change at the atomic level. Iron atoms are originally arranged into crystal structures that change shape when heated; of which, there are two common structures. Depicted in Figure 1 is a body-centered cubic (BCC) crystal structure, which is common in steels at room temperature. Notice that nine total iron atoms make up the unit cell for this arrangement of atoms. Figure 2 depicts a face-centered cubic (FCC) crystal structure. There are 14 total atoms that make up the unit cell for this arrangement of atoms. The FCC transformation occurs when steel is heated above its critical temperature.

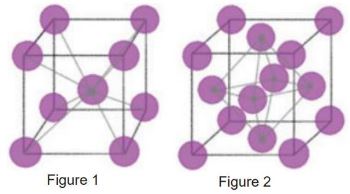
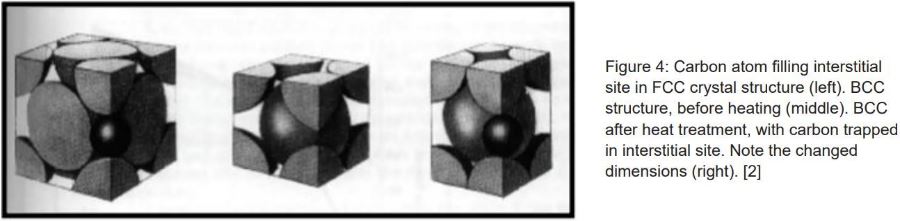
The bonds between iron atoms are relaxed from their BCC state, and transformed into the FCC structure. The important thing to note is the effect of the increased atoms in the lattice. With more atoms, there are more interstitial sites that allow alloying elements to bond with iron and move into these lattices. One such element is carbon, a primary element for hardening steel. Because of the increased amount of interstitial sites that fit carbon, carbon atoms move more freely around iron at elevated temperatures. With greater chance to interrupt geometry of the crystals, steel becomes less ductile, resulting in an increase in strength. To increase the amount of carbon in iron (carburizing), the metal is typically placed in an atmosphere with an elevated carbon level to diffuse additional carbon into the surface.
 Simply heating these steels with an increased carbon atmosphere is not enough to keep it trapped in these lattices to increase hardness. Slow cooling will allow the carbon to diffuse back out, as the structure slowly changes back from FCC to BCC. To counteract this, several different quenchants can be used to cool the material quickly. The quenching allows a quick change of environment for a steel, from high to low temperatures, undergoing heat treatment. It acts to trap carbon and other elements in the middle as there is not enough time for diffusion out of the steel before a change in crystal structure. With these trapped carbon atoms in the crystal structure, we have an altered BCC structure known as martensite.
Simply heating these steels with an increased carbon atmosphere is not enough to keep it trapped in these lattices to increase hardness. Slow cooling will allow the carbon to diffuse back out, as the structure slowly changes back from FCC to BCC. To counteract this, several different quenchants can be used to cool the material quickly. The quenching allows a quick change of environment for a steel, from high to low temperatures, undergoing heat treatment. It acts to trap carbon and other elements in the middle as there is not enough time for diffusion out of the steel before a change in crystal structure. With these trapped carbon atoms in the crystal structure, we have an altered BCC structure known as martensite.
Hardenability
Not every steel reacts the same. Chemical composition can vary greatly between the different grades of steel. Certain alloying elements can greatly increase the hardenability of steels such as nickel (Ni), chromium (Cr) and molybdenum (Mo). Hardenability is not how hard a material is. Hardenability directly relates to the ability of a metal to form martensite and martensistic structure upon quenching, which points to how well hardness can be achieved. Ni, Cr and Mo additions, as well as higher carbon, allow more martensite to form, thus the metal is more “hardenable.” High hardenability is the ability of a metal to transform into the martensite throughout the whole part, not just high hardness at the surface.
Have more questions about heat treating contact us at (319) 232-5221 or fill out our Quick Contact form.
Sources
[1] http://www.gsa.org.au/resources/factites/factitesIron.pdf
[2] http://www.ce.berkeley.edu/~paulmont/CE60New/review1.pdf
Download the PDF version here.